Molecular biology
From CSBLwiki
(Difference between revisions)
(→protocols) |
|||
| Line 10: | Line 10: | ||
===protocols=== | ===protocols=== | ||
| - | + | *[[PCR primer|PCR]] - General rules for primer design | |
| - | + | *[[Genomic DNA extraction]] | |
| - | + | *Competent cell preparation [[media:competentcell.pdf|BSGC protocol (PDF)]] | |
| - | + | *Electrocompetent cell [[media:ElectrocompetentEcoli.pdf|Protocol (PDF)]] | |
| - | + | *LIC cloning introduction [[media:LICnovagen.pdf|LIC introduction]] - Ligation Independent Cloning | |
| - | + | **LIC cloning protocol (Berkeley Structural Genomics Center) | |
| - | + | ***[http://www.strgen.org/protocols/lic-vector-prep.pdf Vector preparation] | |
| - | + | ***[http://www.strgen.org/protocols/lic-insert-prep.pdf Insert preparation] | |
| - | + | ***[http://www.strgen.org/protocols/lic-rxn-transf.pdf LIC transformation]: | |
| - | + | ****mix insert and vector in appropriate amounts | |
| - | + | ****incubate 5~30 min(20 min) at room temperature | |
| - | + | ****do transformation in E. coli (recommend to use electro-competent cells) | |
| - | + | *[http://www.ncbe.reading.ac.uk/ncbe/protocols/ NCBE practical protocols]: very good illustration for undergraduate students | |
| - | + | *Conjugation | |
| - | + | **Donor: <i>E. coli</i> [http://www.atcc.org/ATCCAdvancedCatalogSearch/ProductDetails/tabid/452/Default.aspx?ATCCNum=47055&Template=hosts S17-1] | |
==Tools== | ==Tools== | ||
Revision as of 08:32, 21 July 2009
Contents |
Reagents & Protocols
recipes
- Chemicals
- Commonly used reagents and equipment in molecular biology (PDF)
- Concentration of antibiotics commonly used
- For DNA extraction (to remove some protein contaminants & inactivate enzymes): Phenol:Chloroform=1:1(v:v) solution
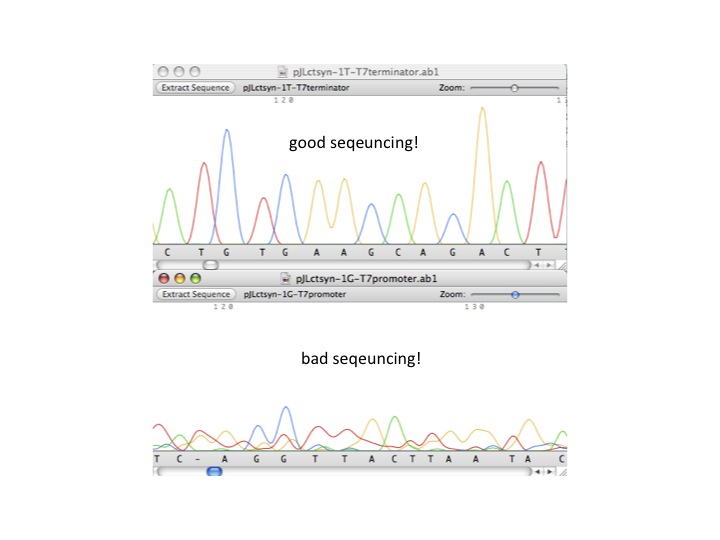
- DNA sequencing check list
- quality score
- good sequencing result vs. bad sequencing result (see below example)
protocols
- PCR - General rules for primer design
- Genomic DNA extraction
- Competent cell preparation BSGC protocol (PDF)
- Electrocompetent cell Protocol (PDF)
- LIC cloning introduction LIC introduction - Ligation Independent Cloning
- LIC cloning protocol (Berkeley Structural Genomics Center)
- Vector preparation
- Insert preparation
- LIC transformation:
- mix insert and vector in appropriate amounts
- incubate 5~30 min(20 min) at room temperature
- do transformation in E. coli (recommend to use electro-competent cells)
- LIC cloning protocol (Berkeley Structural Genomics Center)
- NCBE practical protocols: very good illustration for undergraduate students
- Conjugation
- Donor: E. coli S17-1
Tools
webtools
softwares
- NIH Image: Gel analysis software
- Serial Cloner: DNA sequence management (PCR, restriction maps, etc.)
- BioX (Mac OS X only)
- Biological sequence editor Bioedit
- Jmol